Find Your Perfect Fit: Best Rotator Cuff Sleeping Braces for Pain Relief
February 9, 2026
Compress Your Way to Comfort: Simple Steps for Hot and Cold Packs
February 11, 2026How long has the Intracept procedure been around: 5 Wins!
Why Understanding the Intracept Procedure’s History Matters
If you’re wondering how long has the intracept procedure been around, here’s the quick answer:
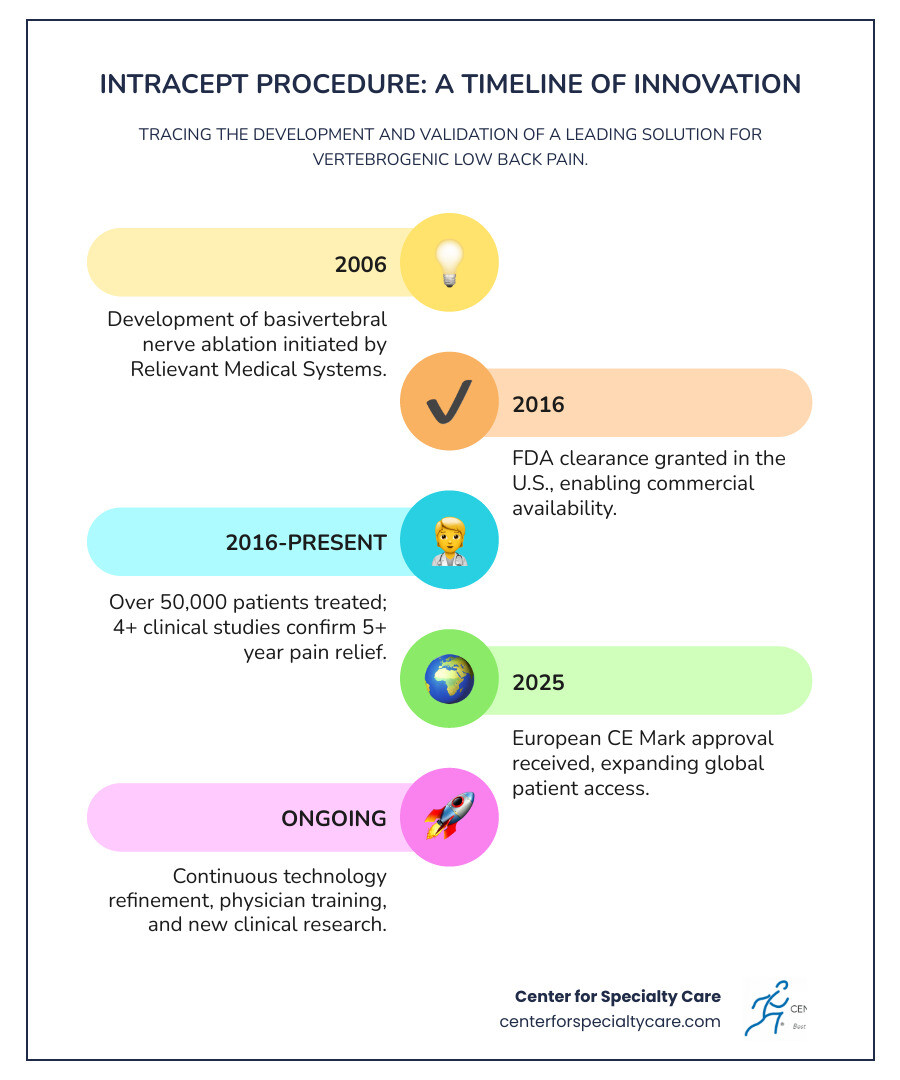
- Development began: 2006 by Relievant Medical Systems
- FDA clearance: 2016 in the United States
- Over 50,000 patients treated across the U.S. as of 2025
- European approval: CE mark received in 2025
- Clinical studies: At least 4 major trials including 2 randomized controlled trials with 5+ year follow-up data
Chronic low back pain affects millions of people every day. For many, conservative treatments like medication, physical therapy, and injections simply don’t provide lasting relief. That’s where newer minimally invasive options come in—but understanding how long has the intracept procedure been around helps patients feel more confident about choosing a treatment with proven results.
The Intracept procedure targets a specific type of chronic low back pain called vertebrogenic pain. This pain originates from damage to the vertebral endplates and the basivertebral nerve that runs through each vertebra. By using radiofrequency energy to disrupt these pain signals, the procedure offers long-term relief without major surgery or implants.
Since its FDA clearance in 2016, the procedure has helped over 50,000 patients across the United States. It’s backed by rigorous clinical research, including randomized controlled trials showing durable results lasting five years or longer. While the technology started development back in 2006, it has been continuously refined and improved—most recently with Boston Scientific’s acquisition and ongoing clinical trials.
I’m Dr. Corey Welchlin, founder of the Center for Specialty Care in Fairmont, Minnesota, and I’ve spent over 30 years helping patients find relief from musculoskeletal pain. Understanding how long has the intracept procedure been around helps me guide patients toward evidence-based treatments that have stood the test of time. Let’s walk through the complete timeline together.
Key terms for how long has the intracept procedure been around:
The Origins: When Was the Intracept Procedure First Developed?
The story of the Intracept procedure begins over a decade ago, with a dedicated team at Relievant Medical Systems. We often marvel at how far medical technology has come, and the Intracept procedure is a prime example of innovation driven by the persistent challenge of chronic low back pain.
The initial development of this groundbreaking treatment began in 2006. Relievant Medical Systems recognized that a significant portion of chronic low back pain patients weren’t finding relief from traditional therapies because the root cause of their pain was often misidentified or untreatable with existing methods. They set out to create a targeted solution.
The core idea behind the Intracept procedure is to address what we call “vertebrogenic pain.” This is a specific type of back pain that originates from damage to the vertebral endplates—the top and bottom surfaces of the vertebrae that connect to the spinal discs. When these endplates are damaged, often due to degenerative changes, they can become inflamed and hypersensitive.
Crucially, the basivertebral nerve (BVN) transmits pain signals from these damaged endplates. The BVN is a sensory nerve that branches throughout the vertebral body, including the endplates. Relievant Medical Systems understood that if they could safely and effectively ablate, or deactivate, this nerve, they could interrupt the pain signals at their source.
Diagnosis of vertebrogenic pain typically involves identifying specific changes on an MRI, known as “Modic changes.” These changes indicate inflammation and damage within the vertebral bone marrow adjacent to the endplates. Before the Intracept procedure, patients with Modic changes often had limited treatment options if conservative care failed. The development of Intracept offered a new pathway for relief.

Understanding the intricate workings of the spine, from its bony structures to its complex nervous system, is vital in treating back pain effectively. We dig deeper into this topic in our guide on Unlocking the Secrets of Spinal Anatomy: A Comprehensive Guide. The precise targeting of the basivertebral nerve, identified through careful anatomical study and imaging, is what makes the Intracept procedure so innovative.
The Journey to Patients: Regulatory Approval and Commercial Availability
Once a medical device is developed, it starts on a rigorous journey through clinical trials and regulatory approvals before it can reach patients. This process ensures safety and efficacy, and for the Intracept procedure, it took several years of dedicated research and validation.
The significant milestone for patients in the United States was the FDA clearance in 2016. This clearance marked the procedure as safe and effective for treating chronic low back pain caused by vertebrogenic factors. Following this crucial approval, the Intracept procedure began its commercial availability, slowly but steadily becoming an option for patients struggling with persistent pain.
More recently, Boston Scientific, a global leader in medical technology, acquired Relievant Medsystems, Inc. in November 2023. This acquisition signifies a major commitment to advancing the Intracept procedure and expanding its reach. Boston Scientific’s resources and expertise are expected to further refine the technology, increase physician training, and improve patient access.
The procedure has also gained international recognition. In July 2025, Boston Scientific announced that the Intracept™ Intraosseous Nerve Ablation System received the CE mark, signaling its introduction and availability in Europe. While our focus here at Center for Specialty Care is on serving our local communities in Fairmont, MN, Estherville, IA, and surrounding areas, this European approval underscores the global acceptance and validation of the Intracept procedure as a viable treatment option. You can read more about this development via this external resource: Intracept™ Intraosseous Nerve Ablation System for vertebrogenic low back pain receives CE mark.
The journey doesn’t end with initial approval. Medical procedures constantly evolve, and the Intracept procedure is no exception. Ongoing research, such as the IMPROVE Study, continues to gather data and explore the procedure’s benefits and applications.

When did the Intracept procedure become available in the U.S.?
For patients suffering from chronic low back pain in the United States, the Intracept procedure became available after receiving FDA clearance in 2016. This was a pivotal moment, as it introduced a new, targeted approach to treating vertebrogenic pain that had previously been difficult to manage.
Since 2016, the procedure has been increasingly adopted by pain management specialists and orthopedic surgeons across the country. Its outpatient nature and minimally invasive profile have made it an attractive option for both patients and providers. Here at Center for Specialty Care, we’ve seen the positive impact this procedure can have on our patients’ lives, providing a pathway to relief when other treatments have failed.
We believe in offering a comprehensive approach to Pain Management, and the Intracept procedure has become an important tool in our arsenal for addressing chronic back pain.
How has the procedure evolved since its inception?
The evolution of the Intracept procedure since its initial development in 2006 has been marked by significant milestones and continuous refinement.
Initially, the focus was on proving its fundamental concept—that ablating the basivertebral nerve could effectively treat vertebrogenic pain. This involved meticulous research, device design, and early clinical trials to establish safety and efficacy.
A major step in its evolution was the acquisition of Relievant Medsystems by Boston Scientific in late 2023. This transition brought the Intracept technology under the umbrella of a larger, well-established medical device company with extensive resources for research, development, and market penetration. This means continued investment in improving the procedure and making it more widely available.
The technology itself has seen refinements in the tools used, making the procedure more precise and efficient for physicians. Alongside technological advancements, there has been a growing emphasis on physician training and education. As more practitioners become proficient in the technique, its adoption rate naturally increases.
Furthermore, the commitment to ongoing clinical research, such as the IMPROVE Study (NCT06827262), demonstrates a continued dedication to understanding and optimizing the Intracept procedure. These studies help to gather more data on long-term outcomes, refine patient selection criteria, and explore potential new applications or improvements. If you’re interested in the cutting-edge research surrounding this procedure, you can Learn more about the IMPROVE study.
The evolution has been from a novel concept to a proven, commercially available, and continuously improving solution for a specific type of chronic low back pain.
How Long Has the Intracept Procedure Been Around and What Does the Research Say?
When considering any medical procedure, especially one designed to alleviate chronic pain, patients rightly ask about its track record. How long has the intracept procedure been around is not just a question of its age, but of the depth and breadth of the scientific evidence supporting it.
The Intracept procedure has been rigorously studied since its development began in 2006. The clinical evidence supporting its effectiveness is robust, including at least four major clinical studies. Crucially, this includes two randomized controlled trials, which are considered the gold standard in medical research. These trials have provided long-term follow-up data, extending to five years and beyond, demonstrating a strong safety profile and consistently meaningful clinical improvements for patients.
These studies have shown that the Intracept procedure offers durable and consistent results, providing patients with significant pain relief and improved function. This long-term data is essential because chronic pain often requires lasting solutions, not just temporary fixes.
The presence of Modic changes on an MRI is a key indicator for the Intracept procedure, as these changes signify the type of vertebral endplate damage that leads to vertebrogenic pain. The clinical studies have focused on patients presenting with these specific MRI findings, ensuring that the research directly applies to the intended patient population.
While many of the external links provided in the research refer to cardiac interventions (which are not relevant to the Intracept procedure), we can draw upon the principles of rigorous scientific inquiry that govern all medical advancements. Just as advancements in interventional cardiology are published in journals like the International Journal of Spine Surgery December 2022 (though this specific link refers to global burden of low back pain and not Intracept specific studies, it highlights the importance of peer-reviewed research in validating treatments), the Intracept procedure’s data has been published in reputable medical literature, providing a foundation of trust and reliability.
How many patients have undergone the Intracept procedure to date?
The adoption rate of the Intracept procedure speaks volumes about its efficacy and the trust it has garnered within the medical community and among patients. As of June 2025, the Intracept procedure has already transformed the lives of more than 50,000 patients across the United States.
This significant number reflects a growing recognition among physicians and patients that the Intracept procedure offers a valuable and effective solution for vertebrogenic low back pain. Each of these 50,000+ patients represents an individual who, after trying various conservative treatments without success, found lasting relief through this innovative approach.
The increasing number of patients successfully treated also contributes to a deeper understanding of the procedure’s real-world effectiveness and safety across a diverse patient population. It reinforces the confidence that we, as healthcare providers at Center for Specialty Care, have in offering this option to our patients in Fairmont, MN, Estherville, IA, and surrounding communities.
What do clinical studies show about its effectiveness?
The clinical studies on the Intracept procedure paint a very positive picture regarding its effectiveness, particularly its ability to provide durable pain relief. Here’s what we’ve learned:
- Meaningful Clinical Improvements: Studies have consistently shown that patients experience significant reductions in pain and improvements in their overall back function. For example, clinical trials report a success rate of 69%, with 64% of patients experiencing a greater than 50% reduction in pain after 12 months. In a multicenter randomized trial, 75.6% of actively treated patients reported meaningful improvement just three months after the treatment compared to a placebo group.
- Durable Results: One of the most compelling aspects of the Intracept procedure is the longevity of its benefits. The rigorous studies include long-term follow-up data, showing that these meaningful improvements are sustained for five years or longer after a single procedure. This is a crucial factor for anyone dealing with chronic pain, as it offers the hope of long-lasting relief rather than a temporary fix.
- Strong Safety Profile: Alongside effectiveness, safety is paramount. The comprehensive clinical trials have also demonstrated a strong safety profile for the Intracept procedure.
- Reduced Need for Other Treatments: By effectively addressing the source of vertebrogenic pain, many patients find that they no longer need to rely as heavily on other pain management strategies, such as injections or ongoing physical therapy, ultimately improving their quality of life.
The evidence is clear: the Intracept procedure offers a highly effective and long-lasting solution for carefully selected patients with vertebrogenic low back pain. For those who have exhausted other options, it represents a significant step forward in reclaiming their lives from chronic discomfort. We are committed to helping our patients find lasting relief for their Back & Spine conditions, and the Intracept procedure is a testament to that commitment.
Understanding Candidacy for the Intracept Procedure Today
Knowing how long has the intracept procedure been around and its proven track record is important, but just as crucial is understanding who is the right candidate for this treatment. Not everyone with low back pain will benefit from the Intracept procedure. We at Center for Specialty Care pride ourselves on personalized care, ensuring each patient receives the most appropriate treatment for their unique situation.
The Intracept™ Intraosseous Nerve Ablation System has specific indications for use. Generally, you might be a candidate if you meet the following criteria:
- Chronic Low Back Pain Duration: You must have experienced chronic low back pain for at least six months. This ensures that the pain is not acute or temporary.
- Failed Conservative Care: You have tried and failed to find significant relief from conservative treatments for at least six months. These typically include physical therapy, medications (over-the-counter and prescription), and injections.
- Modic Changes on MRI: Your MRI scans show features consistent with Modic changes (Type 1 or Type 2). These changes indicate damage and inflammation at the vertebral endplates, which is the anatomical source of vertebrogenic pain that the Intracept procedure targets.
However, it’s equally important to understand who is not a candidate for the Intracept procedure. Contraindications are in place to ensure patient safety and optimize outcomes. You would likely not be a candidate if you:
- Are pregnant or planning to become pregnant.
- Have weakened cardiac (heart) or pulmonary (lung) function.
- Have an active implanted electronic medical device, such as a pacemaker or defibrillator.
- Are diagnosed with a systemic or local infection.
- Have an anatomical condition that could be unintentionally damaged during the procedure.
- Are skeletally immature (generally under 18 years of age).
The Intracept procedure is a minimally invasive option, meaning it doesn’t involve large incisions or extensive spinal reconstruction. This makes it an appealing choice for many, but careful patient selection remains key to its success. We conduct thorough evaluations, including a detailed medical history, physical examination, and review of imaging, to determine if the Intracept procedure is the right path for you.
To learn more about various spinal issues and their treatments, you can explore our page on Common Spinal Conditions: Causes, Symptoms, and Treatment Options.
Frequently Asked Questions about the Intracept Procedure
When considering a new treatment, it’s natural to have questions. We want to ensure you feel fully informed and comfortable with every step of your care journey. Here are some of the most common questions we hear about the Intracept procedure:
Is the Intracept procedure considered a major surgery?
No, the Intracept procedure is definitively not considered a major open surgery. It is a minimally invasive treatment. Unlike traditional open back surgery, which often involves large incisions, muscle dissection, and sometimes implants like screws or rods, the Intracept procedure is performed through a small incision, typically less than half an inch.
We perform the Intracept procedure in an outpatient setting, meaning you can typically go home the same day. There are no implants left in your body after the procedure, and the goal is to disrupt the pain signals at their source without altering the structural mechanics of your spine. This minimally invasive approach generally leads to less tissue damage, a quicker recovery, and fewer risks compared to major spinal surgeries.
For more information on surgical options, both major and minimally invasive, you can visit our Surgery page.
How long does it take to recover from the Intracept procedure?
One of the significant advantages of the Intracept procedure’s minimally invasive nature is its relatively short recovery period. While individual experiences can vary, most patients experience a full recovery within one to two weeks post-procedure.
Here’s a general idea of what to expect:
- Immediately After: You will be monitored as the sedation wears off. You’ll need someone to drive you home. You might experience some mild soreness, bruising, or swelling at the incision site, which can usually be managed with over-the-counter pain relievers.
- First Few Days: We generally recommend resting as much as possible for about 24-48 hours. You should avoid strenuous activities like heavy lifting or intense exercise for a few days to allow the area to heal. Most patients can resume light normal activities fairly quickly. Some individuals even return to work or school as early as two or three days after the procedure, depending on the nature of their work.
- First Few Weeks: A follow-up appointment is typically scheduled two to four weeks after the procedure. This allows us to check on your healing progress and assess your pain relief. Most patients report significant pain relief within two to four weeks after the procedure.
Patience is a virtue, especially when your body is healing. Listen to your body and follow our post-procedure instructions carefully to ensure the best possible outcome.
Are the results of the Intracept procedure permanent?
Yes, the results of the Intracept procedure are designed to be long-lasting, providing durable pain relief. The procedure involves the ablation (deactivation) of the basivertebral nerve. When this nerve is ablated, it permanently stops transmitting pain signals from the damaged vertebral endplates to the brain.
Clinical studies have demonstrated the longevity of these results. Patients in rigorous trials have shown sustained, significant pain reduction for five years or longer after a single Intracept procedure. While the ablation of the specific targeted nerve is permanent, it’s important to understand that the spine is a complex structure, and other pain sources or new degenerative changes could potentially develop over time. However, for the vertebrogenic pain targeted by the Intracept procedure, the relief is considered durable and long-term.
The permanence of the nerve ablation is a key differentiator of the Intracept procedure, offering a solution that goes beyond temporary symptom management.
A Well-Established Solution for Chronic Back Pain
We’ve explored the journey of the Intracept procedure, from its inception by Relievant Medical Systems in 2006 to its FDA clearance in 2016, and its subsequent adoption by over 50,000 patients across the U.S. as of 2025. We’ve also seen how it continues to evolve under Boston Scientific’s stewardship, with ongoing research like the IMPROVE study further solidifying its place in modern pain management.
The question of how long has the intracept procedure been around has a clear answer: it has a well-established history, backed by robust clinical data, including long-term randomized controlled trials. This isn’t a fleeting trend; it’s a proven solution for a specific type of chronic low back pain.
At Center for Specialty Care, serving our communities in Fairmont, MN, Estherville, IA, Buffalo Center, IA, and St. James, MN, we believe in offering treatments with a proven track record. Our commitment to 100% patient satisfaction, personalized care, and quick appointment availability means we thoroughly evaluate each patient to determine if the Intracept procedure is the right, evidence-based option for them.
If you’ve been suffering from chronic low back pain for at least six months, have tried conservative care without success, and your MRI shows Modic changes, the Intracept procedure might be the answer you’ve been searching for. Don’t let chronic pain dictate your life any longer.
We invite you to learn more and explore whether this well-established treatment can bring you the lasting relief you deserve.